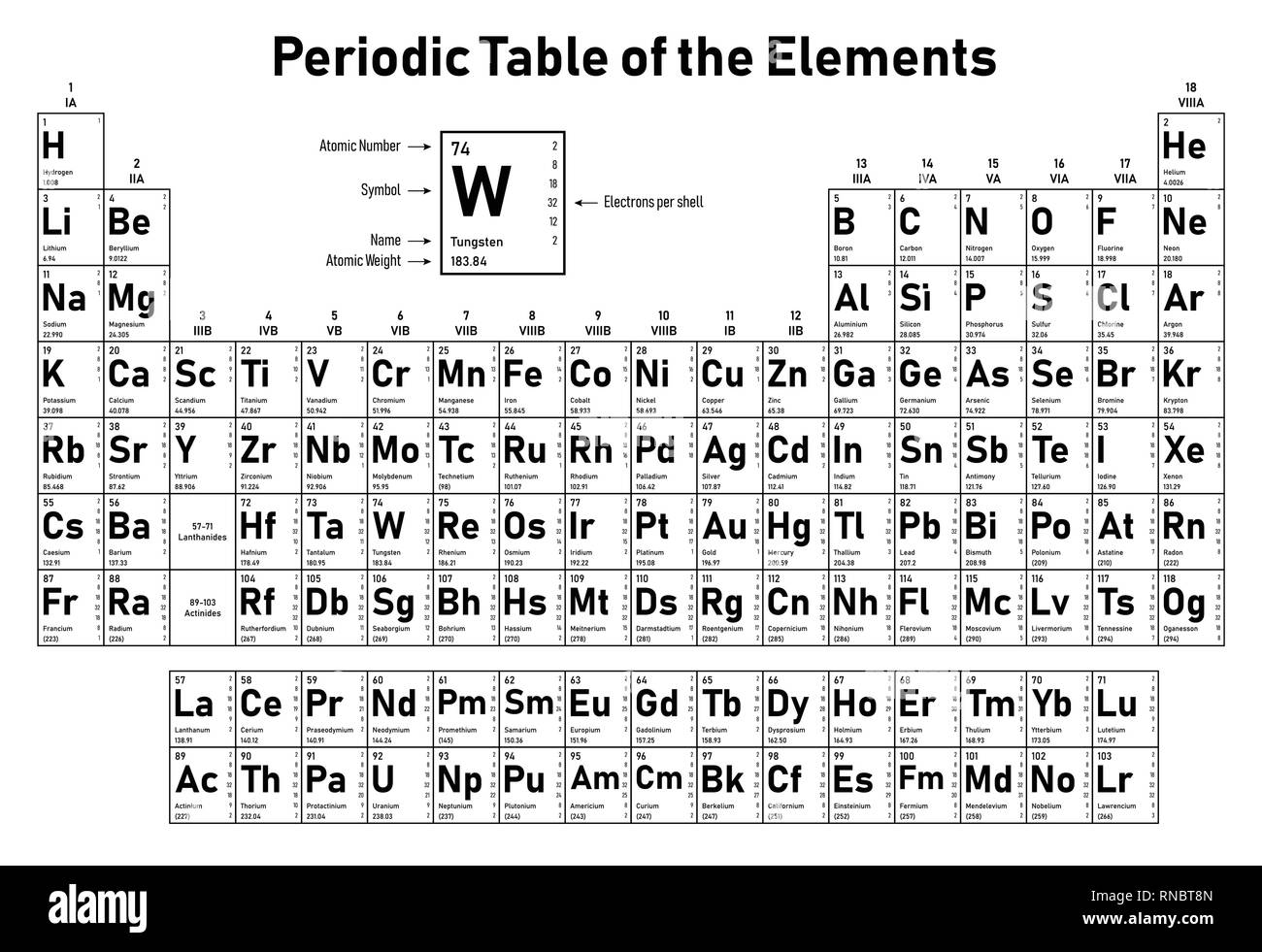
IUPAC also publishes abridged values, rounded to five significant figures. The "(2)" indicates the uncertainty in the last digit shown, to read 4.002 602 ☐.000 002. Typically, such a value is, for example helium: A r°(He) = 4.002 602(2). Of the 118 known chemical elements, 80 have stable isotopes and 84 have this Earth-environment based value. This range is the rationale for the interval notation given for some standard atomic weight values. Standard atomic weight averages such values to the range of atomic weights that a chemist might expect to derive from many random samples from Earth. Non-standardized atomic weights of an element are specific to sources and samples, such as the atomic weight of carbon in a particular bone from a particular archeological site. The definition specifies the use of samples from many representative sources from the Earth, so that the value can widely be used as "the" atomic weight for substances as they are encountered in reality-for example, in pharmaceuticals and scientific research. The standard atomic weight of each chemical element is determined and published by the Commission on Isotopic Abundances and Atomic Weights (CIAAW) of the International Union of Pure and Applied Chemistry (IUPAC) based on natural, stable, terrestrial sources of the element. It can be converted into a measure of mass (with dimension M) by multiplying it with the dalton, also known as the atomic mass constant.Īmong various variants of the notion of atomic weight ( A r, also known as relative atomic mass) used by scientists, the standard atomic weight ( A r°) is the most common and practical. īecause relative isotopic masses are dimensionless quantities, this weighted mean is also dimensionless. The standard atomic weight of a chemical element (symbol A r°(E) for element "E") is the weighted arithmetic mean of the relative isotopic masses of all isotopes of that element weighted by each isotope's abundance on Earth. The standard atomic weight ( A r°(Cu)) for copper is the average, weighted by their natural abundance, and then divided by the atomic mass constant m u. We created a timeline of the history of the periodic table.Not to be confused with Atomic mass. 
The Periodic Table has constantly been improved and developed over the past 200 years, but in 1869 Dimitri Mendeleev finished the first version of the periodic table as we know it today, by arranging the elements by atomic mass and leaving spaces open for the elements that were not yet discovered. 
Who invented the Periodic Table of Elements? The periodic table also gives us an idea of what the characteristics of an element might be and help us predict how an element might react based on in which group it is located. The Periodic Table of Elements can be used as an assisting tool in chemical calculations, when a specification of an element is needed it is easily found in the Periodic Table. How is the Periodic Table of Elements used? The table lists all the elements that are currently known (118), in descending order of the number of protons that are present, in a single atom of the element. The Periodic table of elements is a tool, developed by scientists over hundreds of years. List of all the elements and their properties:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
